

| Lanreotide Basic information |
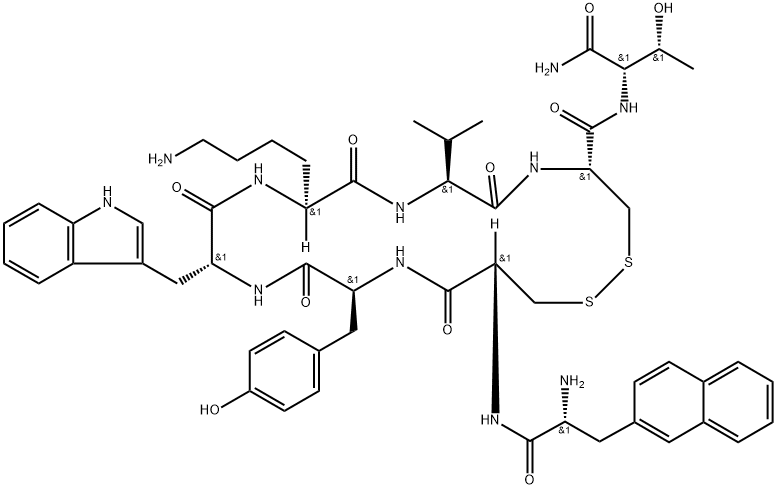
| Product Name: | Lanreotide |
| Synonyms: | LANREOTIDE;AUTOGEL;BETA-(2-NAPHTHYL)-D-ALA-CYS-TYR-D-TRP-LYS-VAL-CYS-THR AMIDE;BIM-23014;ANGIOPEPTIN;H-D-2-NAL-CYS-TYR-D-TRP-LYS-VAL-CYS-THR-NH2;H-D-2-NAL-CYS-TYR-D-TRP-LYS-VAL-CYS-THR-NH2, (DISULFIDE BOND);IPSTYL |
| CAS: | 108736-35-2 |
| MF: | C54H69N11O10S2 |
| MW: | 1096.32 |
| EINECS: | 689-178-4 |
| Shelf life | 2 years |
| Source | Synthetic |
 |
|
| Lanreotide Chemical Properties |
| Boiling point | 1508.2±65.0 °C(Predicted) |
| density | 1.40±0.1 g/cm3(Predicted) |
| storage temp. | Keep in dark place,Sealed in dry,Store in freezer, under -20°C |
| pka | 9.90±0.15(Predicted) |
| Lanreotide Usage And Synthesis |
| Chemical Properties | White to off-white lyophilised powder |
| Description | Lanreotide is indicated for the long-term treatment of acromegalic patients who have had an inadequate response to surgery and/orradiotherapy, or for whom surgery and/or radiotherapy is not an option. The goal of treatment in acromegaly is to reduce (GH) and insuli growth factor-1 levels to normal. |
| Function | Treatment of acromegaly Treatment of certain tumors Radiolabeling in Nuclear Medicine Imaging Managing Bleeding Esophageal Varices Obesity Treatment of Gastrointestinal Fistula |
