

| Butylated Hydroxytoluene Basic information |
| Product Name: | Butylated Hydroxytoluene(p21) |
| Synonyms: | BHT (BAGS);BHT FCC/NF;BHT,GRANULAR,FCC;BHT,GRANULAR,TECHNICAL;BUTYLATEDHYDROXYTOLUENE,GRANULAR,NF;(Z)-retro-αretro-Methyl-αButylated hydroxytoluene Manufacturer |
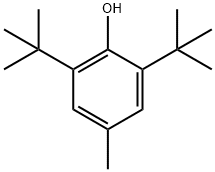
| CAS: | 128-37-0 |
| MF: | C15H24O |
| MW: | 220.35 |
| EINECS: | 204-881-4 |
| Shelf life | 2 years |
| Source | Synthetic |
 |
|
| Butylated Hydroxytoluene Usage And Synthesis |
| Description | Butylated hydroxytoluene is a synthetic phenolic compound mainly used as an antioxidant and preservative in the food industry. It is used to prevent the lipid oxidation in oils and fat-containing foods.Butylated Hydroxytoluene toxicity is generally considered as being low.Since Butylated Hydroxytoluene is used in many near consumer products population wide exposure is expected. |
| Chemical Properties | Butylated hydroxytoluene is white or light yellow crystal. Butylated Hydroxytoluene has a melting point of 71°C, a boiling point of 265°C, a relative density of 1.048 (20/4°C), and a refractive index of 1.4859 (75°C). Solubility of Butylated Hydroxytoluene at normal temperature: methanol 25, ethanol 25-26, isopropanol 30, mineral oil 30, acetone 40, petroleum ether 50, benzene 40, lard (40-50°C ) 40-50, corn oil and soybean oil 40-50. Butylated Hydroxytoluene is insoluble in water, 10NaOH solution, glycerol, and propylene glycol. Butylated Hydroxytoluene is odorless, odorless with good thermal stability. |
| Application from Literature | The applications of butylated hydroxytoluene (BHT) have been reported as following [1-9]: • Butylated hydroxytoluene metabolites causing DNA strand breaks in cultured cells and DNA breaks between nucleosomes (a typical feature of apoptosis), which result in relieving inflammation. • Inhibiting secretion, aggregation, and protein phosphorylation caused by protein kinase C activators at the process of the pre-incubation of aspirin-treated platelets. • Inhibiting liver cancer formation induced by aflatoxin B1. • As Michael receptor, butylated hydroxytoluene can react with uninucleophiles and proteins. • Reaction of 2, 6-di-tert-butyl-4-methylphenol with fluorine (II) - benzophenone dianion complex. • Food additive 2, 6-di-tert-butyl-4-methylphenol can promote acute lung toxicity and tumor growth in mice. • Butylated hydroxytoluene can be used to prepare organoaluminum compound methylaluminum bis (2, 6-di-tert-butyl-4-alkylphenol oxide). |
| Uses | Butylated hydroxytoluene has wide application, such as flavors, fragrances, biochemical reagents-other chemical reagents, chemical raw materials, organic chemical raw materials, biochemical, inorganic salts, antioxidants, food additives, feed additives, feed storage additives, aromatic hydrocarbons, bulk drugs and so on. As a phenolic antioxidant, butylated hydroxytoluene can inhibit lipid peroxidation and exhibit electrophilic quinone methyl ether toxicity mediated by oxidative metabolism. The BHT metabolites, 6-tert-butyl-2- [2 ′-(2′-hydroxymethyl) -propyl] -4-methylphenol, may cause lung damage in mice and promote tumor growth. |
