

| Somatostatin Information |
| Product Name: | Somatostatin |
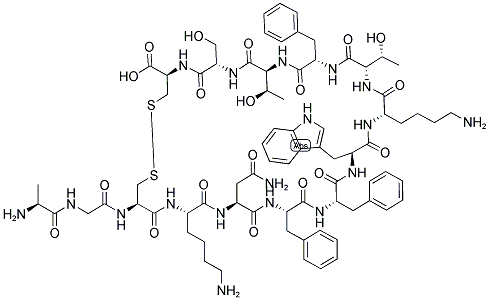
| Synonyms: | panhibin;M.W. 1637.80 C76H104N18O19S2;CCRIS 3629;Somatostatin;Anti-Somatostatin, N-Terminal antibody produced in rabbit;SOMATOSTATIN [GIF];SOMATOSTATIN (HUMAN, OVINE, PORCINE, RAT, MOUSE);SOMATOSTATIN (HUMAN, OVINE, PORCINE, RAT, MOUSE) 2ACOH 6H2O |
| CAS: | 51110-01-1 |
| MF: | C76H104N18O19S2 |
| MW: | 1637.88 |
| EINECS: | 256-969-7 |
| Product Categories: | Peptide;Somatostatin receptor |
 |
|
| Somatostatin Chemical Properties |
| Storage temp. | −20°C |
| Solubility | H2O: 1 mg/mL |
| Form | powder |
| Water Solubility | Soluble in water (0.3 mg/ml) |
| Somatostatin Side effects And Description |
Clinical implications |
Somatostatinoma is a malignant tumor that arises from transformed D cells in the pancreatic islets or duodenum. Somatostatinomas are associated with malabsorption, diabetes mellitus, steatorrhea, and cholelithiasis. In gastroenteropancreatic tumors, high levels of SS receptor expression have been found, and specifically designed analogs are used for tumor imaging and radiotherapy. SS deficiency causes persistent Helicobacter pylori infection in the patient with chronic gastritis. |
Description |
Originally isolated from hypothalamic tissue, somatostatin is characterized as an inhibitor of growth hormone (GH) release. The structure was determined in 1971. Subsequent investigations led to the recognition that somatostatin also was released from the pancreas and has a role of inhibiting the secretion of both insulin and glucagon. A total of five somatostatin receptor subtypes have been characterized and cloned (sst1 to sst5). Subtype sst4 is associated with the inhibition of insulin release, and an sst4-selective inhibitor has been reported. The somatostatin analogue SOM230 has exhibited selectivity for sst1, sst2, sst3, and sst5 in rats and effectively decreased plasma GH and insulin-like growth factor-1 (IGF-1) levels by 75% without significant effects on insulin or glucagon. Another analogue, PT R3173, with selectivity for recombinant human somatostatin receptor (hsst2, hsst4, hsst5) was substantially more effective in inhibiting GH secretion compared to glucagon and insulin release in rats. |
Indications |
Somatostatin (or somatotropin release–inhibiting factor [SRIF]) occurs primarily as a 14–amino acid peptide, although a 28–amino acid form also exists.As with the other hypothalamic peptides, it is formed by proteolytic cleavage of a larger precursor. Somatostatin, originally isolated from the hypothalamus, is also in many other locations, including the cerebral cortex, brainstem, spinal cord, gut, urinary system, and skin. Somatostatin inhibits the secretion of many substances in addition to growth hormone. |
Clinical Use |
Somatostatin has a very brief half-life in serum and is not useful clinically.An 8–amino acid analogue with 2 D-amino acids substituted for the naturally occurring L-amino acids is more stable, and monthly injections of a depot form of this analogue (octreotide, Sandostatin LAR) have several uses. Long-acting octreotide is used to treat acromegaly, as described earlier. It is also used to counteract unpleasant effects caused by overproduction of secreted bioactive substances produced by neuroendocrine tumors, including hyperinsulinemia from insulinomas and secretions from carcinoid tumors that cause severe diarrhea. Octreotide may also control severe diarrhea associated with AIDS that has not responded to other treatments. |
Side effects |
Somatostatin analogues (SSA) are a common treatment for some forms of neuroendocrine tumours (NETs). Somatostatin analogues are usually well tolerated which means you may not have many side effects. The main side effects are Loss of appetite Feeling sick Feeling bloated Stomach pain Fatigue (tiredness) Increased diarrhoea (this is rare) Soreness at the injection site Uncommon side effects include sinus bradycardia, asthenia, headache,pruritus, decreased libido, increased serum bilirubin, and constipation. Transient side effects, gastrointestinal discomfort and decreased glucose tolerance, usually last only a few weeks after initiation of therapy. The most significant side effect associated with prolonged use of octreotide is formation of gallstones resulting from reduced bile flow. |
| storage | -20°C |
