

| Teriparatide acetate Information |
| Product Name: | Teriparatide acetate |
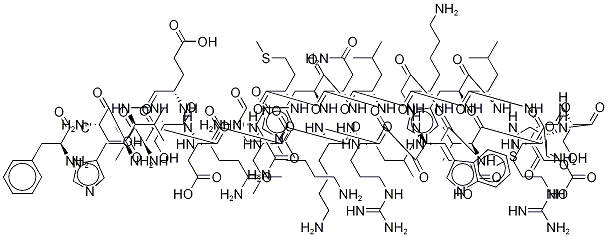
| Synonyms: | PARATHYROID HORMONE HUMAN: FRAGMENT 1-34;PARATHYROID HORMONE (HUMAN, 1-34);PARATHYROID HORMONE (1-34), HUMAN;PTH (1-34) (HUMAN);PTH (HUMAN, 1-34);TERIPARATIDE;Teriparatide acetate;SVSEIQLMHNLGKHLNSMERVEWLRKKLQDVHNF |
| CAS: | 52232-67-4 |
| MF: | C172H278N52O47S2 |
| MW: | 3890.49792 |
| EINECS: | 640-978-1 |
| Mol File: | 52232-67-4.mol |
 |
|
| Teriparatide acetate Chemical Properties |
| Melting point | >205oC (dec.) |
| RTECS | SQ7770000 |
| storage temp. | −20°C |
| solubility | DMSO (Slightly), Water (Slightly) |
| form | powder |
| color | White to Off-White |
| Water Solubility | Soluble to 0.40 mg/ml in water |
| Stability: | Hygroscopic |
| CAS DataBase Reference | 52232-67-4(CAS DataBase Reference) |
| Teriparatide acetate Usage And Synthesis |
| Uses |
A fragment of human parathyroid hormone (hPTH) peptide sequence containing the 34 N-terminal residues of hPTH. This fragment was also found to be an agonist at PTH1 and PTH2 receptors. |
General Description |
Teriparatide is a recombinantform of parathyroid hormone, which is used for the treatmentof osteoporosis in men and postmenopausal women.The N-terminal region possesses 34 amino acids, which areidentical to the biologically active region of the 84-aminoacid sequence of human parathyroid hormone. It has beenshown to act on osteoblasts to stimulate new bone growthand improve bone density. |
Biochem/physiol Actions |
Parathyroid hormone (PTH) is a parathyroid-secreted polypeptide hormone that increases the level of calcium in blood by enhancing calcium mobilization from bone, increasing the calcium:phosphate ratio in the kidney and promoting the absorption of calcium by the intestines. PTH is an anabolic agent that improves osteoblastic bone development. PTH 1-34 induces bone morphogenetic protein (BMP) gene transcription. Active N-terminal fragment of PTH (residues 1–34), has been used as a therapeutic for postmenopausal women with osteoporosis who are at high risk for fracture. |
Clinical Use |
In 2002, the U.S. FDA approved teriparatide for the treatment of postmenopausal osteoporosis in patients who have a high risk of fracture as well as to increase bone mass in men with primary or hypogonadal osteoporosis who have a high risk of fracture. Teriparatide is recombinant human PTH 1-34, the biologically active portion of the endogenously produced preprohormone. Unlike the bisphosphonates, which are classified as bone restorative agents, teriparatide is the first approved bone-forming agent. Bone formation is possible because of the ability of this agent to increase the number of osteoblasts. Although teriparatide enhances the function of both osteoclasts and osteoblasts, the exposure incidence dictates its effect on the skeleton. |
| storage | Store at -20°C |
